Prevention & Treatment
There are two most common methods when it comes to dealing with the malaria disease; one is treatment with medication, which sometimes can be difficult due to the drug resistant nature of the parasites. The other is prevention, which sometimes can be costly and tough to implement, thus, coming up with novel tools and methods is a crucial step in battling the disease. The most recent research has also been targeting the creation of a vaccine for the disease.
Prevention

The burden of malaria
Effective control of malaria has shown many technical and strategic complications. This is due to the nature of the preventive methods that are required to be implemented simultaneously. Long-lasting insecticide-treated bed nets (LLINs) and indoor residual spraying (IRS) often must be used with effective anti-malarial drugs known as malaria prophylaxis to see significant result in effectiveness. However, malaria is a disease that thrives in impoverished areas, and many people who are at risk live in rural areas and lack the sufficient tools and care to these preventive methods [1].
Economics in many African countries often suffer due malaria. Up to 40% of a African nation’s budget are spent on malarial treatment and research each year, and on average a family can spent more than a quarter of its income on treatment and prevention of the disease [2].
Effective control of malaria has shown many technical and strategic complications. This is due to the nature of the preventive methods that are required to be implemented simultaneously. Long-lasting insecticide-treated bed nets (LLINs) and indoor residual spraying (IRS) often must be used with effective anti-malarial drugs known as malaria prophylaxis to see significant result in effectiveness. However, malaria is a disease that thrives in impoverished areas, and many people who are at risk live in rural areas and lack the sufficient tools and care to these preventive methods [1].
Economics in many African countries often suffer due malaria. Up to 40% of a African nation’s budget are spent on malarial treatment and research each year, and on average a family can spent more than a quarter of its income on treatment and prevention of the disease [2].
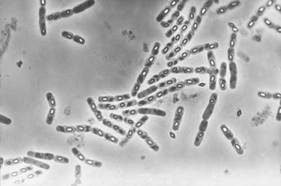
Bti bacteria group
Vector control
Since mosquito is the primary transmitter of the malaria parasite, mosquito eradication is an important step in battling the disease.
The success of malaria control methods in some highly-endemic countries such as Rwanda, Kenya, Gambia, Zanzibar, etc. has reduced the death toll of malaria by 50% or more [3]. The use of LLINs and IRS, together with treatment of uncomplicated Plasmodium falciparum malaria with artemisinin-based combination therapy (ACT), has contributed to the decline in the disease. The effectiveness of these efforts has led to the heightened focus on vector control because historically, the successful eradication of malaria in various parts of the world was primarily done by vector control, which shows that greater importance should be placed in this field, as well as insecticide usage. However, the insecticides has produced many resistant mosquito populations, thus the need for development of new insecticides has high priority. Many novel vector control tools has been developed such as in Kenya, bacillus thuringiensis israelensis (Bti), a bacteria strain that produces toxins to kill mosquito larvae. Other novel tools for the control of the adult mosquito include entomopathogenic fungi, insect-pathogenic viruses, the introduction of genetically engineered mosquitoes and the sterile insect technique (SIT) [4]. Ongoing field-based trials in which the impact of these vector-control tools on public health are needed before they can be adopted to complement the malaria control tools currently in use.
Since mosquito is the primary transmitter of the malaria parasite, mosquito eradication is an important step in battling the disease.
The success of malaria control methods in some highly-endemic countries such as Rwanda, Kenya, Gambia, Zanzibar, etc. has reduced the death toll of malaria by 50% or more [3]. The use of LLINs and IRS, together with treatment of uncomplicated Plasmodium falciparum malaria with artemisinin-based combination therapy (ACT), has contributed to the decline in the disease. The effectiveness of these efforts has led to the heightened focus on vector control because historically, the successful eradication of malaria in various parts of the world was primarily done by vector control, which shows that greater importance should be placed in this field, as well as insecticide usage. However, the insecticides has produced many resistant mosquito populations, thus the need for development of new insecticides has high priority. Many novel vector control tools has been developed such as in Kenya, bacillus thuringiensis israelensis (Bti), a bacteria strain that produces toxins to kill mosquito larvae. Other novel tools for the control of the adult mosquito include entomopathogenic fungi, insect-pathogenic viruses, the introduction of genetically engineered mosquitoes and the sterile insect technique (SIT) [4]. Ongoing field-based trials in which the impact of these vector-control tools on public health are needed before they can be adopted to complement the malaria control tools currently in use.
Treatment
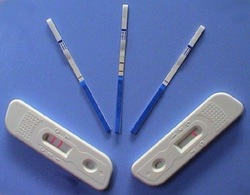
Rapid Diagnostic Test Kit (RDT)
Improving diagnosis
The accuracy of diagnosis is a vital part of good malaria case management and is becoming increasingly important. The proportion of people treated for malaria who have a confirmed diagnosis is low in the African Region compared with other regions of the world [3], with the result that anti-malarials could be used to treat patients without malaria.
Biological diagnostic tools such as rapid diagnostic tests (RDTs) are simple, affordable and give fast results. The introduction of RDTs at the community level needs to be carefully planned, to include transport and storage considerations, local sensitivity testing and establishment of a comprehensive quality assessment/control system. To overcome the likelihood of prescribing anti-malarial medications despite a negative test result poses a significant challenge. The necessity for training, supervision, and follow-up are paramount in achieving a change in perceptions and medical practice.
If managed this process would reduce the unnecessary administration of these drugs and may also help to combat the spread of resistance by ensuring that only malaria patients receive this treatment. This revised approach to malaria diagnosis may also have an impact on patients' perceptions of anti-malarial therapies. Patients will have greater confidence in a therapy if it is shown to work quickly and effectively. Thus, the need for correct initial diagnosis will encourage future compliance [3].
Malaria control slowed due to drug resistance
The progress and expansion of parasitic resistance to anti-malarial mediators is a major obstacle to the success of malaria eradication in certain areas, and has contributed to the resurgence of infection and the increase in malaria-related deaths in recent years [3].
Resistance to almost all commonly used anti-malarials medications has been observed in the most lethal parasite species, P. falciparum [3]. The problem of resistance exists in much of Africa and Southeast Asia; treatment failure rates of up to 80% have been reported for chloroquine, which was formerly the cheapest and most widely available anti-malarial drug.
The half-life of anti-malarials is believed to be an important factor in the development and spread of resistance [5]. It has been suggested that compounds with a longer half-life have a greater tendency to become ineffective due to parasite resistance and exposure to minute concentration of chemicals. Malaria parasites would not be exposed to partially effective drug concentrations if the drugs are eliminated during the two-day life cycle of the asexual parasite. However, it has also been hypothesized that a longer half-life offers prolonged protection against re-infection for a longer period of time, an effect known as "post-treatment prophylaxis” [6]. Unfortunately, what benefits an individual patient may be unfavorable to society if a greater risk of drug resistance develops as more parasites are exposed to sub-therapeutic drug levels.
The accuracy of diagnosis is a vital part of good malaria case management and is becoming increasingly important. The proportion of people treated for malaria who have a confirmed diagnosis is low in the African Region compared with other regions of the world [3], with the result that anti-malarials could be used to treat patients without malaria.
Biological diagnostic tools such as rapid diagnostic tests (RDTs) are simple, affordable and give fast results. The introduction of RDTs at the community level needs to be carefully planned, to include transport and storage considerations, local sensitivity testing and establishment of a comprehensive quality assessment/control system. To overcome the likelihood of prescribing anti-malarial medications despite a negative test result poses a significant challenge. The necessity for training, supervision, and follow-up are paramount in achieving a change in perceptions and medical practice.
If managed this process would reduce the unnecessary administration of these drugs and may also help to combat the spread of resistance by ensuring that only malaria patients receive this treatment. This revised approach to malaria diagnosis may also have an impact on patients' perceptions of anti-malarial therapies. Patients will have greater confidence in a therapy if it is shown to work quickly and effectively. Thus, the need for correct initial diagnosis will encourage future compliance [3].
Malaria control slowed due to drug resistance
The progress and expansion of parasitic resistance to anti-malarial mediators is a major obstacle to the success of malaria eradication in certain areas, and has contributed to the resurgence of infection and the increase in malaria-related deaths in recent years [3].
Resistance to almost all commonly used anti-malarials medications has been observed in the most lethal parasite species, P. falciparum [3]. The problem of resistance exists in much of Africa and Southeast Asia; treatment failure rates of up to 80% have been reported for chloroquine, which was formerly the cheapest and most widely available anti-malarial drug.
The half-life of anti-malarials is believed to be an important factor in the development and spread of resistance [5]. It has been suggested that compounds with a longer half-life have a greater tendency to become ineffective due to parasite resistance and exposure to minute concentration of chemicals. Malaria parasites would not be exposed to partially effective drug concentrations if the drugs are eliminated during the two-day life cycle of the asexual parasite. However, it has also been hypothesized that a longer half-life offers prolonged protection against re-infection for a longer period of time, an effect known as "post-treatment prophylaxis” [6]. Unfortunately, what benefits an individual patient may be unfavorable to society if a greater risk of drug resistance develops as more parasites are exposed to sub-therapeutic drug levels.
Anti-malarial Medication
Medicating effective anti-malarials
Rapid treatment of malaria cases can help reduce the rate of malaria transmission.
Artemisinin derivatives are exceptionally powerful anti-malarials with rapid onset rate, and when administered in combination with anti-malarial drugs with slower elimination rates such as lumefantrine, short courses of treatment (three days) have proved to be highly effective in trials [3]. Combination of anti-malarial medications has shown increased effectiveness and decreases the chance of parasitic resistance.
Artemisinin derivatives can reduce parasite load by a factor of approximately 10,000 per asexual cycle [7], compared with a reduction of 100-fold to 1,000-fold per cycle with most other anti-malarial drugs [3]. They also have gametocidal properties, which could have an important effect on the reoccurrence of malaria. Artemisinin can lower transmission of the infection by inhibiting gametocyte development. Other drug combination is also under clinical testing and trials.
Development of improved pediatric medication
Unfortunately many of the malarial infected patients are infants and young children, thus, appropriate medication for them is vital. However, the administering process presents a particular challenge because it can be difficult, stressful, and time consuming. The drug tablets, often, need to be crushed and mixed with food or drink to ease the process, therefore, possibly reducing the effective dose and the bitter taste can cause spitting and regurgitation. Despite some syrup or drop form exists, the stability and hygiene of the medication cannot be guaranteed. Accurate dosage can also pose complication for families lacking the proper measuring tools.
A new dispersible formulation of Artemether and Lumefantrine tablets, specifically designed for children, has proved in clinical trials to be as safe and effective as the regular tablet formulation. The tablets quickly dissolve in a small amount of fluid and produce a sweet-tasting formulation for ease of administration to infants and children [8].
Rapid treatment of malaria cases can help reduce the rate of malaria transmission.
Artemisinin derivatives are exceptionally powerful anti-malarials with rapid onset rate, and when administered in combination with anti-malarial drugs with slower elimination rates such as lumefantrine, short courses of treatment (three days) have proved to be highly effective in trials [3]. Combination of anti-malarial medications has shown increased effectiveness and decreases the chance of parasitic resistance.
Artemisinin derivatives can reduce parasite load by a factor of approximately 10,000 per asexual cycle [7], compared with a reduction of 100-fold to 1,000-fold per cycle with most other anti-malarial drugs [3]. They also have gametocidal properties, which could have an important effect on the reoccurrence of malaria. Artemisinin can lower transmission of the infection by inhibiting gametocyte development. Other drug combination is also under clinical testing and trials.
Development of improved pediatric medication
Unfortunately many of the malarial infected patients are infants and young children, thus, appropriate medication for them is vital. However, the administering process presents a particular challenge because it can be difficult, stressful, and time consuming. The drug tablets, often, need to be crushed and mixed with food or drink to ease the process, therefore, possibly reducing the effective dose and the bitter taste can cause spitting and regurgitation. Despite some syrup or drop form exists, the stability and hygiene of the medication cannot be guaranteed. Accurate dosage can also pose complication for families lacking the proper measuring tools.
A new dispersible formulation of Artemether and Lumefantrine tablets, specifically designed for children, has proved in clinical trials to be as safe and effective as the regular tablet formulation. The tablets quickly dissolve in a small amount of fluid and produce a sweet-tasting formulation for ease of administration to infants and children [8].
Education is Key
Educating healthcare workers and patients about the prevention and treatment of malaria is another challenge in the management of the disease. One of the greatest difficulties in reducing the toll of malaria is reaching remote communities with poor transport systems, and achieving timely reordering to maintain supplies of medications. Many organizations is working together to local governments to share information on the best practices in their countries and communities including topics such as awareness, training, stock management and health impact measurements [3]. Thus through cooperation, hopefully the impact of malaria can be controlled and eventually eradicated.
Vaccines
A vaccine is a biological agent that improves a person’s immunity to a particular disease. Vaccine typically contains an agent that resembles a disease-causing microorganism, in this case the malarial parasite, and it is often made from one of its surface proteins. The agent stimulates the body's immune system to recognize the agent as foreign, destroy it, and in this manner, the immune system is capable of recognizing the parasite and can more easily destroy it during later encounters.
Complications with a vaccine for malaria
1. Polymorphism and clonal variation in antigens of plasmodium is a complication in creating a general vaccine due to the many species of parasite.
2. Parasite induced immuno-suppression causes the reduced effectiveness of the immune system.
3. Intracellular parasitic spores make it hard for immune system to target.
4. Lack of MS proteins on infected RBCs. (refer to pathophysiology)
5. It is not possible yet to grow the malarial parasite in large quantities.
6. Effectively evaluation methods are still being researched.
7. Malarial parasite is extremely effective in avoiding the host’s immune response.
8. Complications in the conduction of clinical and field trials.
9. The parasite is highly mutable.
10. There are a large number of antigenic variations. For example, MSA- has 8 variants, MSA-2 has 10, and CSP has 6 variants.
Not only are the antigens specific to plasmodium species, they are specific to stage of parasitic development as well.
1. Polymorphism and clonal variation in antigens of plasmodium is a complication in creating a general vaccine due to the many species of parasite.
2. Parasite induced immuno-suppression causes the reduced effectiveness of the immune system.
3. Intracellular parasitic spores make it hard for immune system to target.
4. Lack of MS proteins on infected RBCs. (refer to pathophysiology)
5. It is not possible yet to grow the malarial parasite in large quantities.
6. Effectively evaluation methods are still being researched.
7. Malarial parasite is extremely effective in avoiding the host’s immune response.
8. Complications in the conduction of clinical and field trials.
9. The parasite is highly mutable.
10. There are a large number of antigenic variations. For example, MSA- has 8 variants, MSA-2 has 10, and CSP has 6 variants.
Not only are the antigens specific to plasmodium species, they are specific to stage of parasitic development as well.
Approaches in developing a vaccine
The difficulty of developing a highly effective malaria vaccine has led to the design and assessment of a very wide range of new approaches unparalleled in any other area of infectious disease vaccinology. Malaria is used as a testing ground for many novel new vaccine technologies targeting sporozoites or blood-stage inoculations [10].
Twelve approaches to a malaria vaccine.
1. Sporozoite subunit vaccination, especially with the Cell Surface proteins: e.g. RTS, S in adjuvant.
2. Irradiated sporozoite or genetically attenuated sporozoite immunization either by mosquito bite or using injected purified sporozoites.
3. Immunization with DNA and/or viral vectors to induce T cells against the liver-stage parasites, or to target other life cycle stages.
4. Use of whole blood-stage malaria parasites as immunogens.
5. Use of protein in adjuvant vaccines to reduce the growth rate of blood-stage parasites.
6. Use of protein (or long peptide) in adjuvant vaccines to induce antibody-dependent cellular inhibition (ADCI) of blood-stage parasites.
7. Use of peptide-based vaccines, mainly against blood-stage parasites—e.g. SPf66, PEV3a.
8. Development of anti-disease vaccines based on parasite toxins—e.g. GPI-based.
9. Immunization with parasite adhesion ligands such as PfEMP1.
10. Use of parasite antigens, such as the Var2 protein, preferentially expressed in the placenta to prevent malaria in pregnancy.
11. Immunization with sexual stage parasite antigens as transmission-blocking vaccines.
12. Use of mosquito antigens as transmission-blocking vaccines.
Some pioneering advances to malaria vaccines
1. Development of novel adjuvant formulations inducing exceptional levels of antibody: e.g. AS01.
2. Recombinant particle development: e.g. RTS,S vaccine.
3. First reported clinical trial of DNA vaccination.
4. Large-scale testing of a peptide-based vaccine, SPf66.
5. Discovery and clinical development of prime-boost immunization with vectors.
6. Demonstration of T-cell-mediated protection with sub-unit vaccination.
7. Clinical assessment of recombinant vaccine, multiple antigenic peptide and long peptide vaccines.
8. Development of the concept of community/transmission-blocking immunization, with sexual stage antigens.
9. Development of a pathogen challenge model widely used to guide vaccine development.
10. Development of a regulatory compliant process for whole parasite vaccine bio-manufacture.
The difficulty of developing a highly effective malaria vaccine has led to the design and assessment of a very wide range of new approaches unparalleled in any other area of infectious disease vaccinology. Malaria is used as a testing ground for many novel new vaccine technologies targeting sporozoites or blood-stage inoculations [10].
Twelve approaches to a malaria vaccine.
1. Sporozoite subunit vaccination, especially with the Cell Surface proteins: e.g. RTS, S in adjuvant.
2. Irradiated sporozoite or genetically attenuated sporozoite immunization either by mosquito bite or using injected purified sporozoites.
3. Immunization with DNA and/or viral vectors to induce T cells against the liver-stage parasites, or to target other life cycle stages.
4. Use of whole blood-stage malaria parasites as immunogens.
5. Use of protein in adjuvant vaccines to reduce the growth rate of blood-stage parasites.
6. Use of protein (or long peptide) in adjuvant vaccines to induce antibody-dependent cellular inhibition (ADCI) of blood-stage parasites.
7. Use of peptide-based vaccines, mainly against blood-stage parasites—e.g. SPf66, PEV3a.
8. Development of anti-disease vaccines based on parasite toxins—e.g. GPI-based.
9. Immunization with parasite adhesion ligands such as PfEMP1.
10. Use of parasite antigens, such as the Var2 protein, preferentially expressed in the placenta to prevent malaria in pregnancy.
11. Immunization with sexual stage parasite antigens as transmission-blocking vaccines.
12. Use of mosquito antigens as transmission-blocking vaccines.
Some pioneering advances to malaria vaccines
1. Development of novel adjuvant formulations inducing exceptional levels of antibody: e.g. AS01.
2. Recombinant particle development: e.g. RTS,S vaccine.
3. First reported clinical trial of DNA vaccination.
4. Large-scale testing of a peptide-based vaccine, SPf66.
5. Discovery and clinical development of prime-boost immunization with vectors.
6. Demonstration of T-cell-mediated protection with sub-unit vaccination.
7. Clinical assessment of recombinant vaccine, multiple antigenic peptide and long peptide vaccines.
8. Development of the concept of community/transmission-blocking immunization, with sexual stage antigens.
9. Development of a pathogen challenge model widely used to guide vaccine development.
10. Development of a regulatory compliant process for whole parasite vaccine bio-manufacture.
Vaccines targeting whole parasite
Partly as a response to the limited effectiveness achieved by RTS, S some pharmacologist has been researching to develop a pre-erythrocytic vaccine comprising whole sporozoites. The challenges facing this approach are considerable but the main driver has been the appreciation that irradiated sporozoites delivered by mosquito bite have induced very high levels of protective efficiency, exceeding 90 per cent, though the numbers of volunteers in these trials were very small. Irradiated sporozoites can invade liver cells and develop within them to produce defective schizonts. These express antigens that can induce a protective immune response but the defective schizonts cannot rupture to release the merozoites that would normally invade red blood cells and continue the infection [11].
Vector vaccines
Another major approach to inducing pre-erythrocytic immunity has been to employ vectored vaccines, aiming mainly to induce cellular immunity against the liver-stage of P. falciparum. Irradiated sporozoite-induced immunity in animal models is due to mainly CD8+ T cells and appears to target multiple antigens [11]. Several generations of vectored vaccines have now been assessed clinically in attempts to induce comparable efficacy. However, even in mice, it has been difficult to generate high-level efficacy with vectors encoding single antigens, not least because the levels of T cells required are exceptionally high.
Partly as a response to the limited effectiveness achieved by RTS, S some pharmacologist has been researching to develop a pre-erythrocytic vaccine comprising whole sporozoites. The challenges facing this approach are considerable but the main driver has been the appreciation that irradiated sporozoites delivered by mosquito bite have induced very high levels of protective efficiency, exceeding 90 per cent, though the numbers of volunteers in these trials were very small. Irradiated sporozoites can invade liver cells and develop within them to produce defective schizonts. These express antigens that can induce a protective immune response but the defective schizonts cannot rupture to release the merozoites that would normally invade red blood cells and continue the infection [11].
Vector vaccines
Another major approach to inducing pre-erythrocytic immunity has been to employ vectored vaccines, aiming mainly to induce cellular immunity against the liver-stage of P. falciparum. Irradiated sporozoite-induced immunity in animal models is due to mainly CD8+ T cells and appears to target multiple antigens [11]. Several generations of vectored vaccines have now been assessed clinically in attempts to induce comparable efficacy. However, even in mice, it has been difficult to generate high-level efficacy with vectors encoding single antigens, not least because the levels of T cells required are exceptionally high.
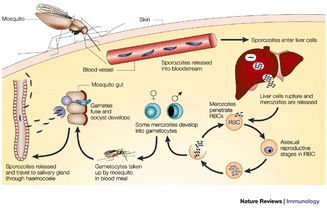
Life Cycle of Malaria Parasite
Blood-stage vaccines
In contrast to major progress in several areas of pre-erythrocytic vaccine development, results with blood-stage vaccines have been more mixed and progress has generally been slower. A number of candidate vaccines have progressed to clinical testing but none has yet achieved good evidence of protective efficacy against clinical malaria. Many of these vaccine candidates are based on just a few antigens, MSP1 and AMA1 in particular, although there are hundreds or perhaps thousands of antigens expressed by blood-stage parasites that might be used in vaccine development. Almost all of these candidate vaccines have been a protein given with a vaccine agent designed to induce protective antibodies. Some approaches have focused on inducing antibodies that impair parasite growth, as can be demonstrated in in vitro assays, whereas others have aimed to induce antibodies which achieve their effect in collaboration with effector cells and which can be measured, with greater difficulty, in a functional assay of ADCI of parasite growth [12].
In contrast to major progress in several areas of pre-erythrocytic vaccine development, results with blood-stage vaccines have been more mixed and progress has generally been slower. A number of candidate vaccines have progressed to clinical testing but none has yet achieved good evidence of protective efficacy against clinical malaria. Many of these vaccine candidates are based on just a few antigens, MSP1 and AMA1 in particular, although there are hundreds or perhaps thousands of antigens expressed by blood-stage parasites that might be used in vaccine development. Almost all of these candidate vaccines have been a protein given with a vaccine agent designed to induce protective antibodies. Some approaches have focused on inducing antibodies that impair parasite growth, as can be demonstrated in in vitro assays, whereas others have aimed to induce antibodies which achieve their effect in collaboration with effector cells and which can be measured, with greater difficulty, in a functional assay of ADCI of parasite growth [12].
Mosquito stage vaccines
The idea of using gametocyte or sexual stage parasite antigens to immunize individuals who might derive no direct benefit but protect their neighbors from becoming infected has been described as altruistic vaccination. Such vaccines would be deployed in such a manner that the whole community would benefit and hence the term ‘community vaccine’ is becoming more popular. Recently, the broader term ‘vaccines that interrupt malaria transmission’ (VIMTs) has been introduced as any pre-erythrocytic or blood-stage vaccine that was highly efficacious could have an effect on transmission [13].
The idea of using gametocyte or sexual stage parasite antigens to immunize individuals who might derive no direct benefit but protect their neighbors from becoming infected has been described as altruistic vaccination. Such vaccines would be deployed in such a manner that the whole community would benefit and hence the term ‘community vaccine’ is becoming more popular. Recently, the broader term ‘vaccines that interrupt malaria transmission’ (VIMTs) has been introduced as any pre-erythrocytic or blood-stage vaccine that was highly efficacious could have an effect on transmission [13].
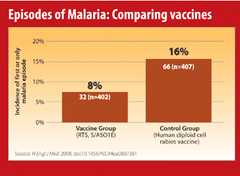
Phase 2 trial of RTS,S/AS01
RTS, S, a possible effective vaccine:
One of the most effective malaria vaccine tested to date is RTS, S, which is a hybrid protein particle in a multi-component vaccine agent named AS01, and it targets the pre-erythrocytic stage of the disease.
In November 2012, findings from a Phase III trial of RTS, S reported that it provided modest protection against both clinical and severe malaria in young infants. The RTS, S vaccine was engineered using genes from the outer protein of Plasmodium falciparum malaria parasite and a portion of a hepatitis B virus and a chemical adjuvant to boost the immune system response. The RTS, S based vaccine formulation had previously been demonstrated to be safe, well tolerated, immunogenic, and to potentially confer partial efficacy in both malaria-naive and -experienced adults as well as children, although further research was considered necessary to improve the effectiveness of the vaccine [10].
One of the most effective malaria vaccine tested to date is RTS, S, which is a hybrid protein particle in a multi-component vaccine agent named AS01, and it targets the pre-erythrocytic stage of the disease.
In November 2012, findings from a Phase III trial of RTS, S reported that it provided modest protection against both clinical and severe malaria in young infants. The RTS, S vaccine was engineered using genes from the outer protein of Plasmodium falciparum malaria parasite and a portion of a hepatitis B virus and a chemical adjuvant to boost the immune system response. The RTS, S based vaccine formulation had previously been demonstrated to be safe, well tolerated, immunogenic, and to potentially confer partial efficacy in both malaria-naive and -experienced adults as well as children, although further research was considered necessary to improve the effectiveness of the vaccine [10].
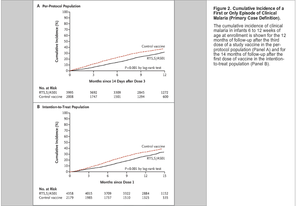
A phase III trial of RTS, S/AS01 Malaria vaccine in African Infants [10]
The candidate malaria vaccine RTS, S/AS01 reduced episodes of both clinical and severe malaria in children 5 to 17 months of age by approximately 50% in an ongoing phase 3 trial. Infants 6 to 12 weeks of age were recruited for the same trial. RTS, S/AS01 or a comparator vaccine were administered to 6537 infants who were 6 to 12 weeks of age at the time of the first vaccination in conjunction with Expanded Program on Immunization (EPI) vaccines in a three-dose monthly schedule.
Vaccine efficacy against the first or only episode of clinical malaria during the 12 months after vaccination, a co-primary end point, was analyzed with the use of Cox regression. Vaccine efficacy against all malaria episodes, vaccine efficacy against severe malaria, safety, and immunogenicity were also assessed.
The results discovered that the incidence of the first or only episode of clinical malaria in the intention-to-treat population during the 14 months after the first dose of vaccine was 0.31 per person-year in the RTS, S/AS01 group and 0.40 per person-year in the control group, for a vaccine efficacy of 30.1%. Vaccine efficacy in the per-protocol population was 31.3%. Vaccine efficacy against severe malaria was 26.0% in the intention-to treat population and 36.6% in the per-protocol population.
Serious adverse events occurred with a similar frequency in the two study groups.
One month after administration of the third dose of RTS, S/AS01, 99.7% of children were positive for anti-circum sporozoite antibodies.
The final conclusions drawn was the RTS, S/AS01 vaccine co-administered with EPI vaccines only provided modest protection against both clinical and severe malaria in young infants.
The candidate malaria vaccine RTS, S/AS01 reduced episodes of both clinical and severe malaria in children 5 to 17 months of age by approximately 50% in an ongoing phase 3 trial. Infants 6 to 12 weeks of age were recruited for the same trial. RTS, S/AS01 or a comparator vaccine were administered to 6537 infants who were 6 to 12 weeks of age at the time of the first vaccination in conjunction with Expanded Program on Immunization (EPI) vaccines in a three-dose monthly schedule.
Vaccine efficacy against the first or only episode of clinical malaria during the 12 months after vaccination, a co-primary end point, was analyzed with the use of Cox regression. Vaccine efficacy against all malaria episodes, vaccine efficacy against severe malaria, safety, and immunogenicity were also assessed.
The results discovered that the incidence of the first or only episode of clinical malaria in the intention-to-treat population during the 14 months after the first dose of vaccine was 0.31 per person-year in the RTS, S/AS01 group and 0.40 per person-year in the control group, for a vaccine efficacy of 30.1%. Vaccine efficacy in the per-protocol population was 31.3%. Vaccine efficacy against severe malaria was 26.0% in the intention-to treat population and 36.6% in the per-protocol population.
Serious adverse events occurred with a similar frequency in the two study groups.
One month after administration of the third dose of RTS, S/AS01, 99.7% of children were positive for anti-circum sporozoite antibodies.
The final conclusions drawn was the RTS, S/AS01 vaccine co-administered with EPI vaccines only provided modest protection against both clinical and severe malaria in young infants.
References
[1] Roll Back Malaria Partnership. Africa Malaria Report - 2003. Chapter 2: Insecticide-treated nets http://rbm.who.int/amd2003/amr2003/pdf/ch2.pdf
[2] Roll Back Malaria Partnership. Africa Malaria Report - 2003. Chapter 6: Resource mobilization and financing http://rbm.who.int/amd2003/amr2003/pdf/ch6.pdf
[3] WHO World Malaria Report 2008 http://www.who.int/malaria/wmr2008
[4] Takken W, Knols BG. Malaria vector control: current and future strategies. Trends Parasitol.2009;25:101–104. doi: 10.1016/j.pt.2008.12.002
[5] White NJ. Antimalarial drug resistance. J Clin Invest. 2004;113:1084–1092.
[6] White NJ. How antimalarial drug resistance affects post-treatment prophylaxis. Malar J.2008;7:9. doi: 10.1186/1475-2875-7-9.
[7] White NJ, van Vugt M, Ezzet F. Clinical pharmacokinetics and pharmacodynamics of artemether-lumefantrine. Clin Pharmacokinet. 1999;37:105–125. doi: 10.2165/00003088-199937020-00002.
[8] Novartis Drug Regulatory Affairs Coartem®/Riamet® Dispersible Basic Prescribing Information. 2009.
[9] Moorthy V. S., et al. 2009. Report of a consultation on the optimization of clinical challenge trials for evaluation of candidate blood stage malaria vaccines, 18–19 March 2009, Bethesda, MD, USA. Vaccine27, 5719–5725 (doi:10.1016/j.vaccine.2009.07.049) doi: 10.1016/j.vaccine.2009.07.049.
[10] "A Phase 3 Trial of RTS,S/AS01 Malaria Vaccine in African Infants". New England Journal of Medicine. 2012.
[11] Hoffman S. L., et al. 2002. Protection of humans against malaria by immunization with radiation-attenuated Plasmodium falciparum sporozoites. J. Infect. Dis. 185, 1155–1164 (doi:10.1086/339409) doi: 10.1086/339409.
[12] Ellis R. D., et al. 2009. A Phase 1 study of the blood-stage malaria vaccine candidate AMA1-C1/Alhydrogel with CPG 7909, using two different formulations and dosing intervals. Vaccine 27, 4104–4109 (doi:10.1016/j.vaccine.2009.04.077) doi: 10.1016/j.vaccine.2009.04.077.
[13] The malERA Consultative Group on Vaccines 2011. A research agenda for malaria eradication: vaccines. PLoS Med 8, e1000398. (doi:10.1371/journal.pmed.1000398) doi: 10.1371/journal.pmed.1000398.
Images:
LLINs: http://images-en.busytrade.com/92556100/Sell-Rectangular-Insecticide-Treated-Mosquito-Net.jpg
RDT kit: http://image.made-in-china.com/2f0j00pBeECbARHauM/HIV-1-2-Rapid-Diagnostic-Test-Kit.jpg
Coartem: http://www.undispatch.com/un-content/uploads/2012/04/Screen-shot-2012-04-25-at-1.02.08-PM.png
Vaccine: http://theintelhub.com/wp-content/uploads/2010/07/vaccine1.jpg
Malaria lifecycle: http://www.nature.com/nri/journal/v1/n2/images/nri1101-117a-f1.gif
Graph: "A Phase 3 Trial of RTS,S/AS01 Malaria Vaccine in African Infants". New England Journal of Medicine. 2012.
[2] Roll Back Malaria Partnership. Africa Malaria Report - 2003. Chapter 6: Resource mobilization and financing http://rbm.who.int/amd2003/amr2003/pdf/ch6.pdf
[3] WHO World Malaria Report 2008 http://www.who.int/malaria/wmr2008
[4] Takken W, Knols BG. Malaria vector control: current and future strategies. Trends Parasitol.2009;25:101–104. doi: 10.1016/j.pt.2008.12.002
[5] White NJ. Antimalarial drug resistance. J Clin Invest. 2004;113:1084–1092.
[6] White NJ. How antimalarial drug resistance affects post-treatment prophylaxis. Malar J.2008;7:9. doi: 10.1186/1475-2875-7-9.
[7] White NJ, van Vugt M, Ezzet F. Clinical pharmacokinetics and pharmacodynamics of artemether-lumefantrine. Clin Pharmacokinet. 1999;37:105–125. doi: 10.2165/00003088-199937020-00002.
[8] Novartis Drug Regulatory Affairs Coartem®/Riamet® Dispersible Basic Prescribing Information. 2009.
[9] Moorthy V. S., et al. 2009. Report of a consultation on the optimization of clinical challenge trials for evaluation of candidate blood stage malaria vaccines, 18–19 March 2009, Bethesda, MD, USA. Vaccine27, 5719–5725 (doi:10.1016/j.vaccine.2009.07.049) doi: 10.1016/j.vaccine.2009.07.049.
[10] "A Phase 3 Trial of RTS,S/AS01 Malaria Vaccine in African Infants". New England Journal of Medicine. 2012.
[11] Hoffman S. L., et al. 2002. Protection of humans against malaria by immunization with radiation-attenuated Plasmodium falciparum sporozoites. J. Infect. Dis. 185, 1155–1164 (doi:10.1086/339409) doi: 10.1086/339409.
[12] Ellis R. D., et al. 2009. A Phase 1 study of the blood-stage malaria vaccine candidate AMA1-C1/Alhydrogel with CPG 7909, using two different formulations and dosing intervals. Vaccine 27, 4104–4109 (doi:10.1016/j.vaccine.2009.04.077) doi: 10.1016/j.vaccine.2009.04.077.
[13] The malERA Consultative Group on Vaccines 2011. A research agenda for malaria eradication: vaccines. PLoS Med 8, e1000398. (doi:10.1371/journal.pmed.1000398) doi: 10.1371/journal.pmed.1000398.
Images:
LLINs: http://images-en.busytrade.com/92556100/Sell-Rectangular-Insecticide-Treated-Mosquito-Net.jpg
RDT kit: http://image.made-in-china.com/2f0j00pBeECbARHauM/HIV-1-2-Rapid-Diagnostic-Test-Kit.jpg
Coartem: http://www.undispatch.com/un-content/uploads/2012/04/Screen-shot-2012-04-25-at-1.02.08-PM.png
Vaccine: http://theintelhub.com/wp-content/uploads/2010/07/vaccine1.jpg
Malaria lifecycle: http://www.nature.com/nri/journal/v1/n2/images/nri1101-117a-f1.gif
Graph: "A Phase 3 Trial of RTS,S/AS01 Malaria Vaccine in African Infants". New England Journal of Medicine. 2012.
